- May 9, 2026
- Genetics
- 0 Comments
Andes Hantavirus: A Comprehensive Guide
History, Transmission, Symptoms, and Advanced Molecular Detection Methods
Published May 2026 • Agam Diagnostics
History of Andes Hantavirus
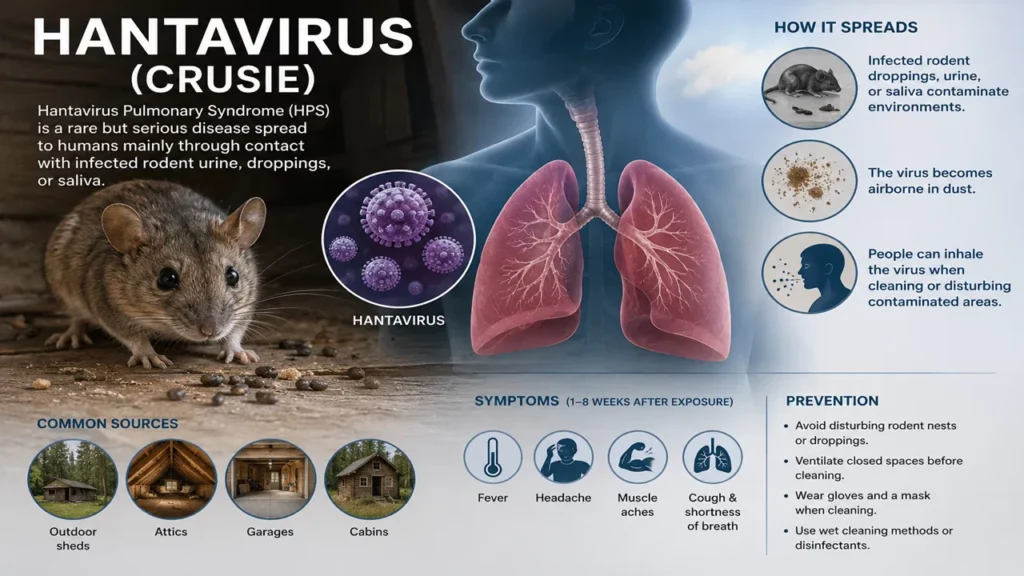
Andes virus (ANDV), a member of the Orthohantavirus genus, was first identified in 1995 in Argentina and Chile following outbreaks of Hantavirus Pulmonary Syndrome (HPS). It is named after the Andes mountain range where the initial cases were reported. The virus gained global attention due to its unique ability among hantaviruses to transmit from person to person, first documented during the 1996 outbreak in El Bolsón, Argentina.
Since then, ANDV has become the primary cause of HPS in South America, with approximately 100–200 cases reported annually, mainly in Argentina and Chile. Recent events, including the 2026 cruise ship outbreak, have highlighted its potential for clustered transmission in confined environments.
Genetic Material and Structure
Andes virus possesses a tri-segmented, negative-sense, single-stranded RNA genome approximately 12.1 kilobases in length:
- S (Small) segment (~1.87 kb): Encodes the nucleoprotein (N) and a non-structural protein that inhibits interferon production.
- M (Medium) segment (~3.67 kb): Encodes a glycoprotein precursor cleaved into Gn and Gc spike proteins responsible for cell attachment and entry.
- L (Large) segment (~6.56 kb): Encodes the RNA-dependent RNA polymerase (RdRp) for genome replication and transcription.
The virus is enveloped with glycoprotein spikes, forming spherical or pleomorphic particles 80–160 nm in diameter. This genetic structure enables persistent infection in rodent hosts while causing severe disease in humans.
Transmission
Primary transmission occurs through contact with infected rodents, particularly the long-tailed pygmy rice rat (Oligoryzomys longicaudatus):
- Inhalation of aerosolized virus from rodent urine, saliva, or feces
- Bites or scratches from infected rodents
- Contaminated food or surfaces
Incubation Period
Median: 18 days
Reported range (CDC)
Symptoms typically appear 1–8 weeks after exposure. Viral RNA can be detectable in blood up to two weeks before symptom onset.
Symptoms of Hantavirus Pulmonary Syndrome (HPS)
Prodromal Phase (3–5 days): Fever, fatigue, muscle aches (especially large muscles), headache, dizziness, chills, nausea, vomiting, diarrhea, abdominal pain.
Cardiopulmonary Phase: Sudden onset of cough, shortness of breath, rapid progression to pulmonary edema, low blood oxygen, cardiogenic shock, and respiratory failure. This phase can develop rapidly within hours.
Fatality Rate
Case fatality rate for Andes virus HPS
Higher than many other hantaviruses. Early intensive care and supportive treatment (oxygen, mechanical ventilation) can improve outcomes. No specific antiviral exists.
Precautionary Measures
- Avoid contact with rodents and their habitats in endemic areas (South America)
- Use rodent-proof storage for food; seal homes and campsites
- Wear N95 masks and gloves when cleaning rodent-infested areas
- Practice rigorous hand hygiene
- For person-to-person risk: Maintain distance, avoid sharing utensils/drinks, use PPE in healthcare settings
- Monitor symptoms for up to 42 days after potential exposure
Detection Methods at Agam Diagnostics
Early and accurate diagnosis is critical. Agam Diagnostics offers state-of-the-art molecular testing:
Recommended Lab Tests
- RT-PCR (Real-Time Polymerase Chain Reaction): Gold standard for detecting viral RNA in blood during early infection. Highly sensitive and specific for Andes virus.
- IgM/IgG ELISA Serology: Detects antibodies. IgM appears early; paired sera confirm rising titers.
- Next-Generation Sequencing (NGS): For strain confirmation and outbreak tracking.
Contact Agam Diagnostics for specialized hantavirus panels and emergency testing protocols.
Request Testing InformationDisclaimer: This information is for educational purposes. Consult healthcare professionals for medical advice. Agam Diagnostics provides advanced diagnostic solutions for infectious diseases.
© 2026 Agam Diagnostics • All Rights Reserved
